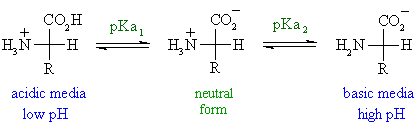
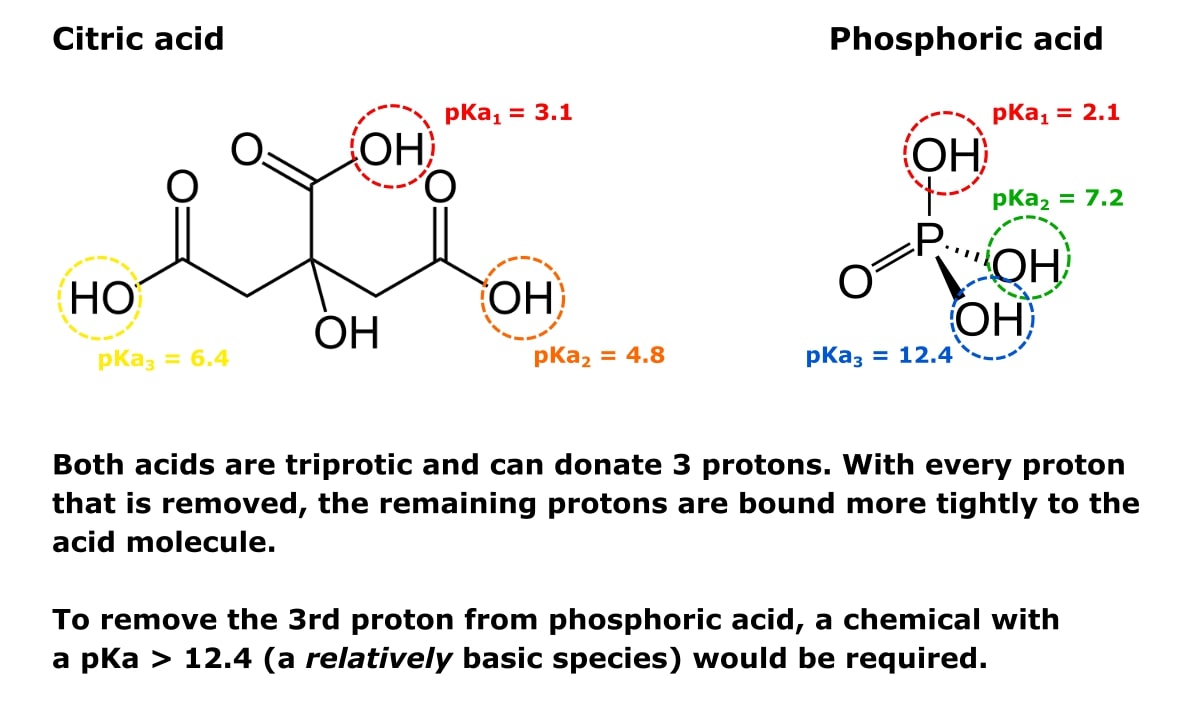
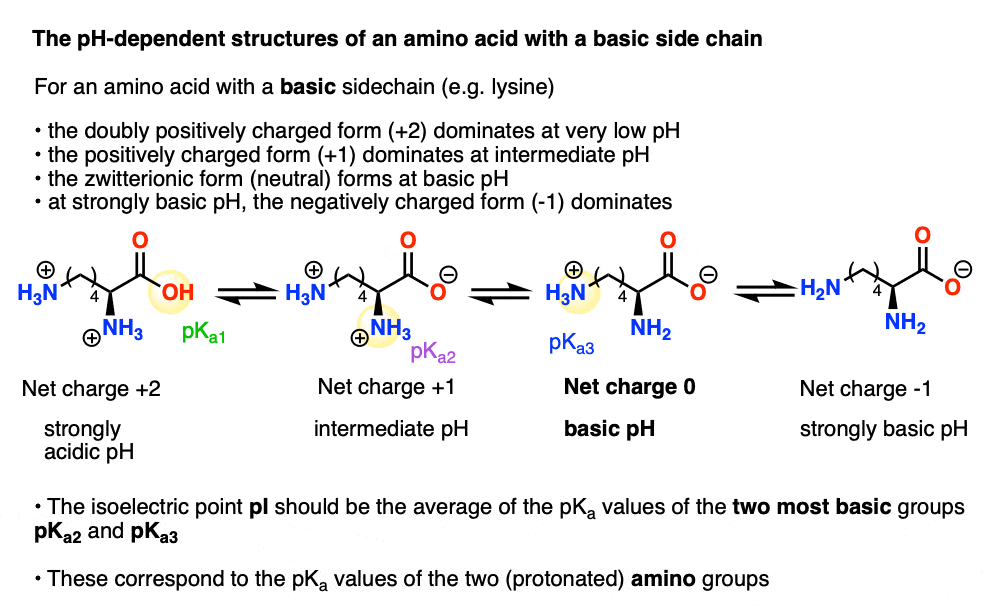
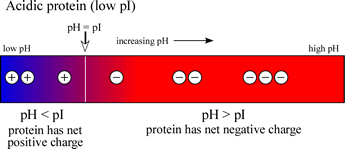
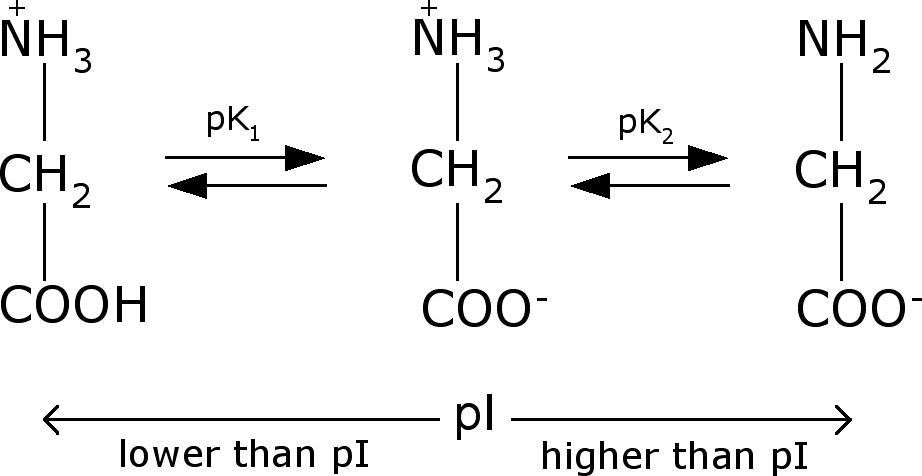
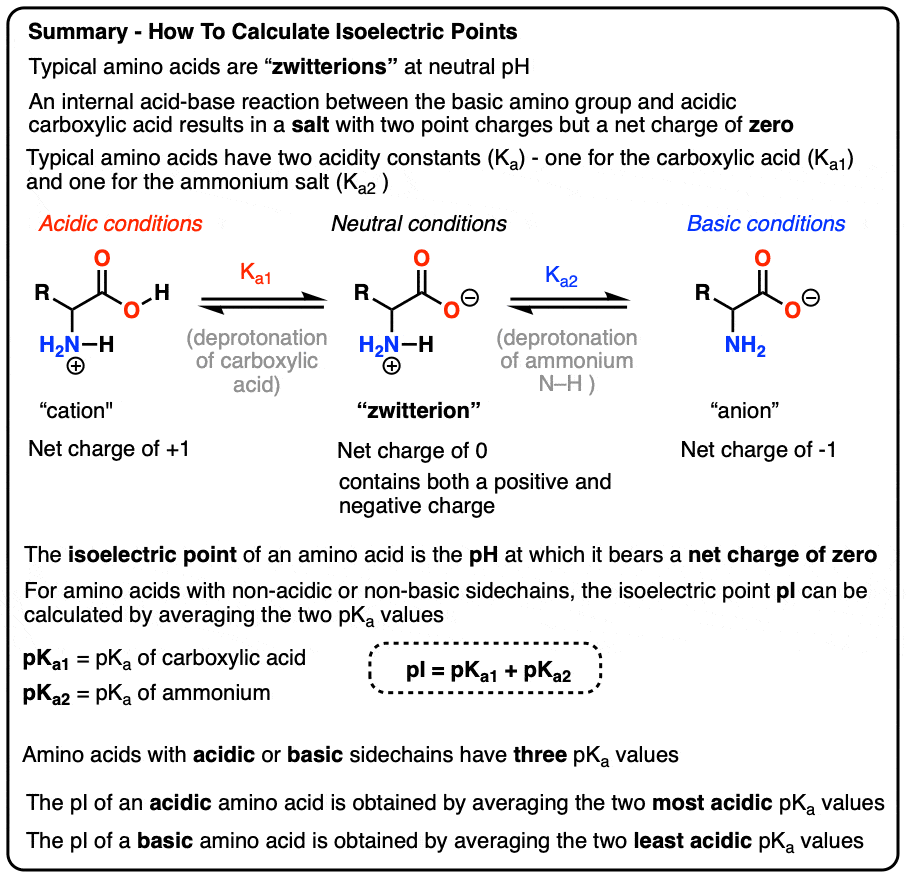
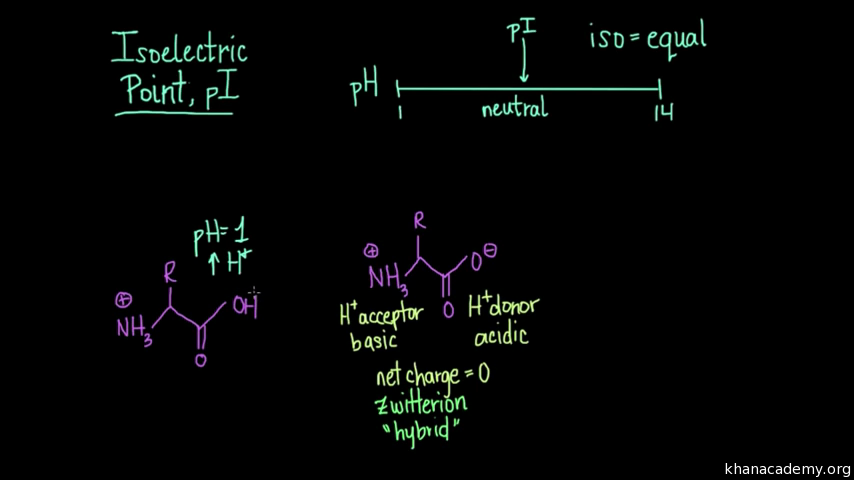
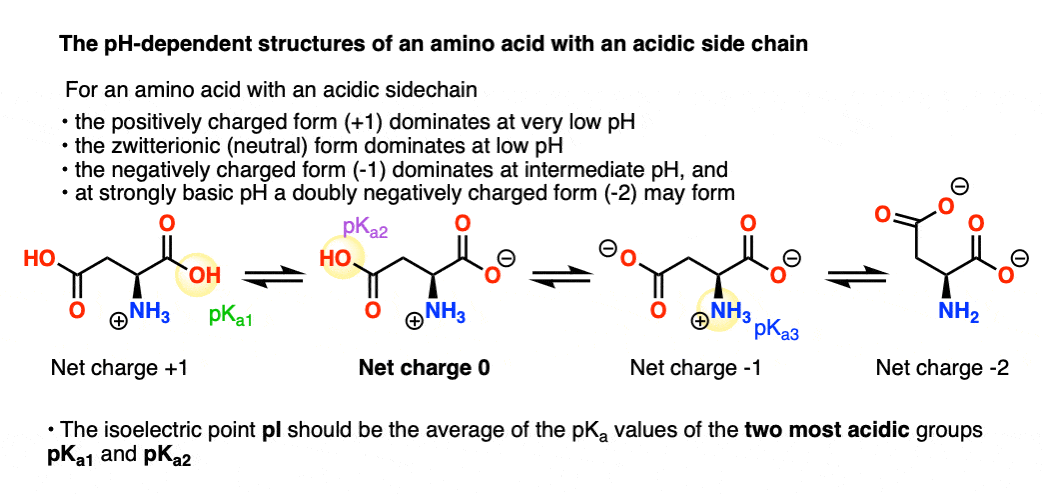
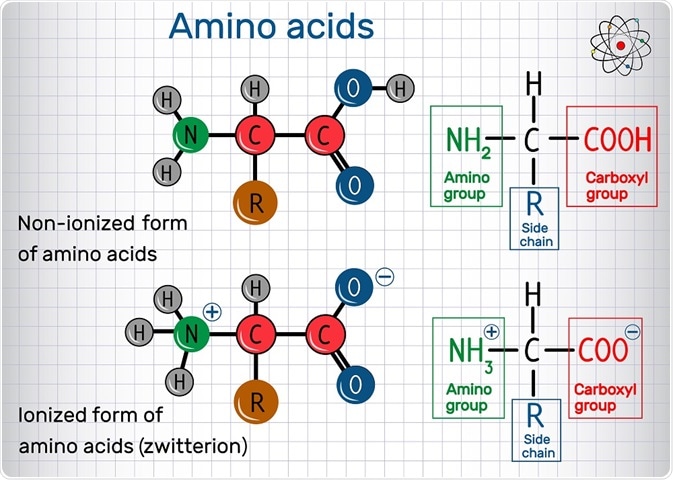
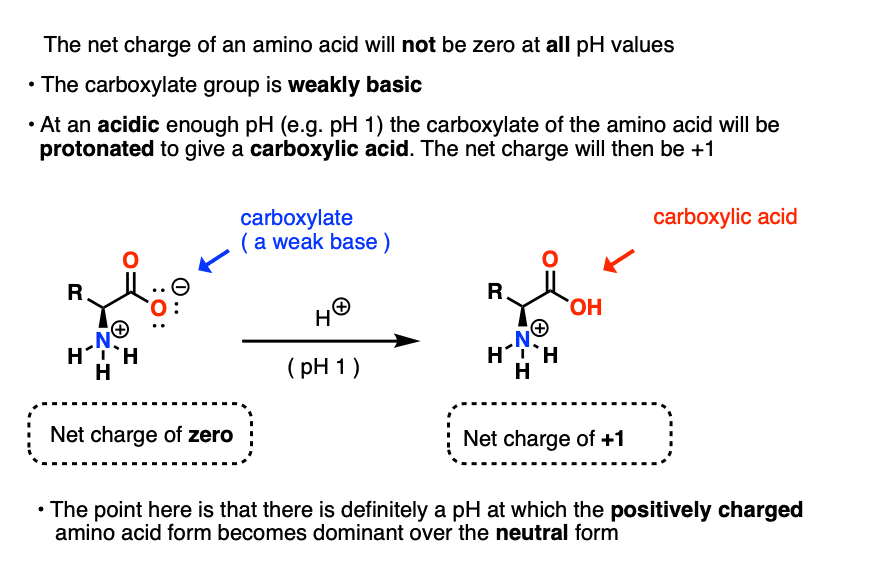
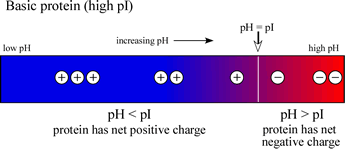
Define isoelectric point (pI)? How does it affect net charge on an amino acid? Calculate (show the calculations) the pI for histidine, and aspartic acid. pK values for these amino acids can

Localization-specific distributions of protein pI in human proteome are governed by local pH and membrane charge | BMC Molecular and Cell Biology | Full Text